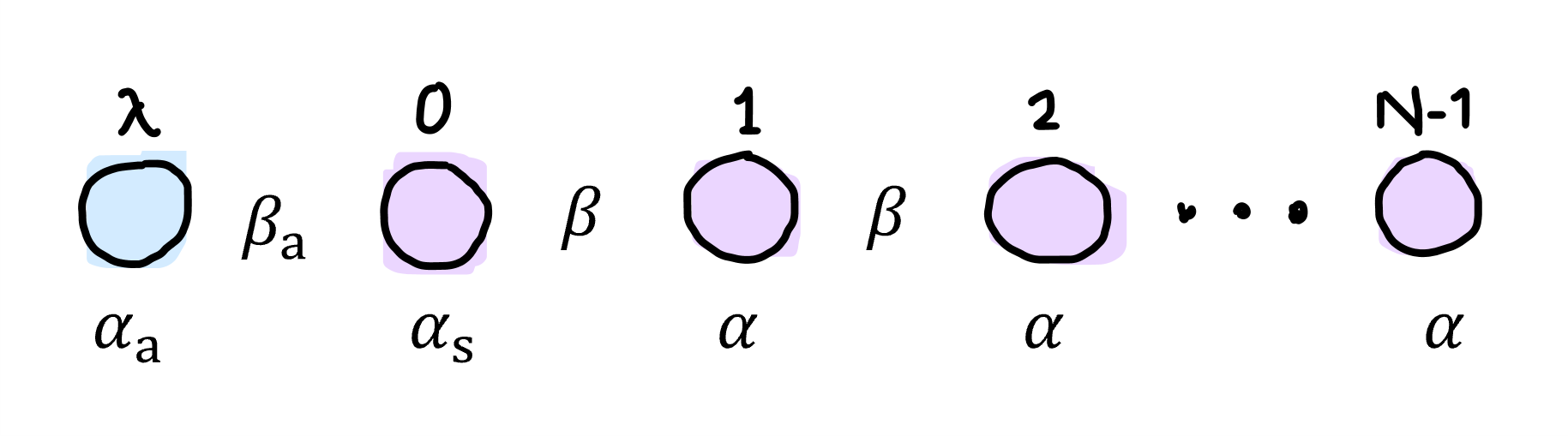
Each atom has an atomic orbital with for the adsorbate and the substrate. We can write the total system wavefunction (molecular orbital) as an LCAO, where indicates different energy levels of the molecular orbitals:
Or in bra-ket notation:
where .
Each molecular orbital satisfies the 1-electron Schrödinger equation
By inserting and multiplying by , we get the Schrödinger equation in terms of matrix elements:
If we assume orthonormal atomic orbitals, . We also write the Hamiltonian in the tight-binding approximation: $$ H =
which transforms our Schrödinger equation into
This equation has three ‘boundary conditions’:
For :
For :
For :
Now we take the Ansatz . Actually, this ‘plane wave Ansatz’ is a common Ansatz for a wave function in periodic systems, and corresponds to Bloch’s theorem. The book provides a derivation of this Ansatz from scratch. By inserting this Ansatz, the Schrödinger equation can then be written as
This defines the energy band. Hence, is related to the band width.
We can also express the LCAO coefficient at any point in terms of the plane waves and the coefficient at (actually not entirely clear to me how using positive and negative waves should be motivated here),
or by re-expressing some parameters in terms of others
Now we apply the boundary conditions. gives
and with the cosine/sine sum rule this yields
which implies that .
The other two boundary conditions can also be applied. Through some algebra (including some cosine/sine sum rules), one gets an ‘eigenvalue equation’:
where in the derivation process the following parameters were defined:
the chemisorption parameter
the surface parameter
the chemisorption (adbond) parameter .
Solutions¶
Let’s consider solutions for . Then the solutions to the eigenvalue problem satisfy either
(if ), or
The first equation gives one solution and the second one gives solutions.
The solutions are visible in the graph below, where the purple lines are two possible values of (1 and -1),
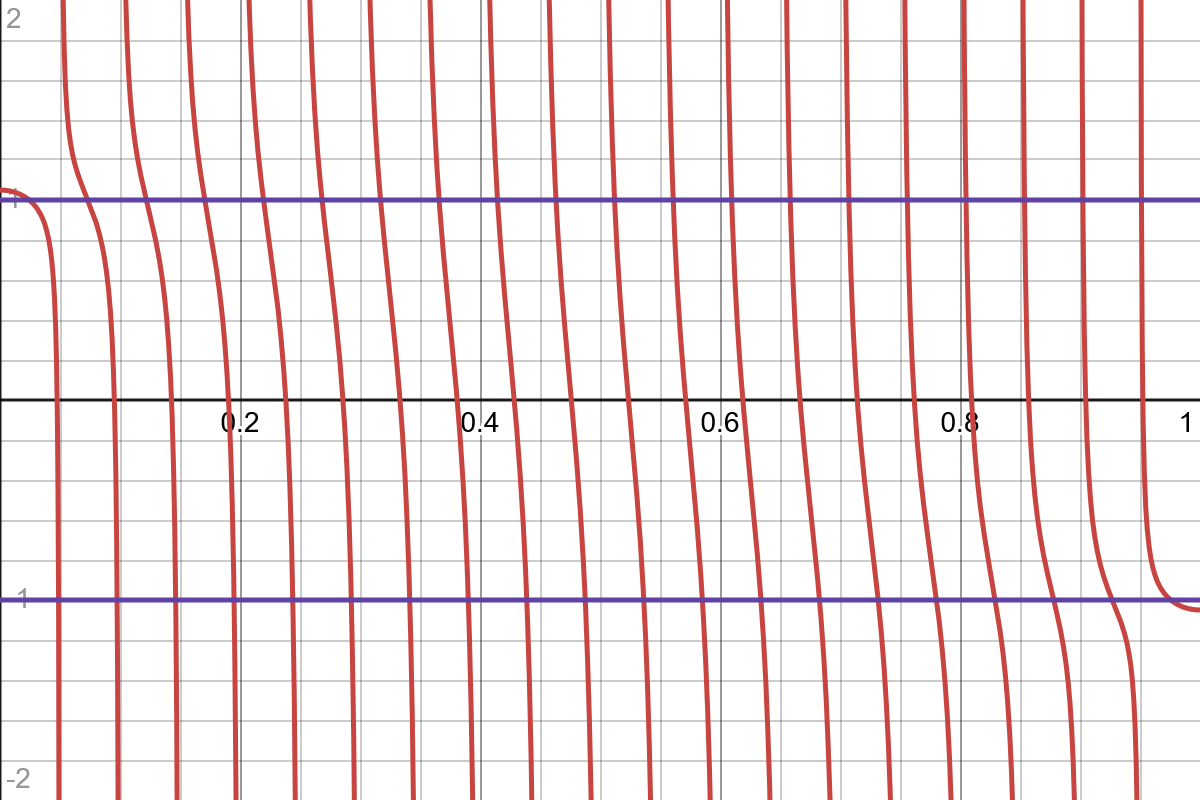
But when becomes larger than 1 or smaller than -1, a solution disappears -- at least it does on the real number line. We can consider solutions for complex , where (it does not make sense to include , because then we get real solutions; I think the reason to not include negative is because it will give duplicate solutions). We can then write . However, the energy must be real, so , which gives . Consequently is either 1 or -1, so and already describe the complete space of solutions, higher just gives duplicates.
So overall we get two possible additional solutions by considering complex ,
The positive one lies above the bulk band, and we call them P-states. The negative one lies below the bulk band, and we call them N-states.
In conclusion, there are atoms, each with one electron, so there are molecular orbital states, i.e. solutions for :
real solutions in the bulk band.
1 solution for the adatom
1 solution for the surface The latter two may be real (in bulk band), or complex (P-states and N-states).
We now show that bulk band states are delocalized and P/N-states are localized. Find the corresponding graph on Desmos.
Bulk band states¶
Oscillates in with constant period. So delocalized.
P-states¶
Large and if has a positive imaginary part :
If :
which is a decaying wavefunction.
Inserting the large- approximation for the wavefunction in the ‘eigenvalue equation’, we get
By setting we can solve this equation graphically; we get a function
and we find the intersections with
The graphical solution is shown below:
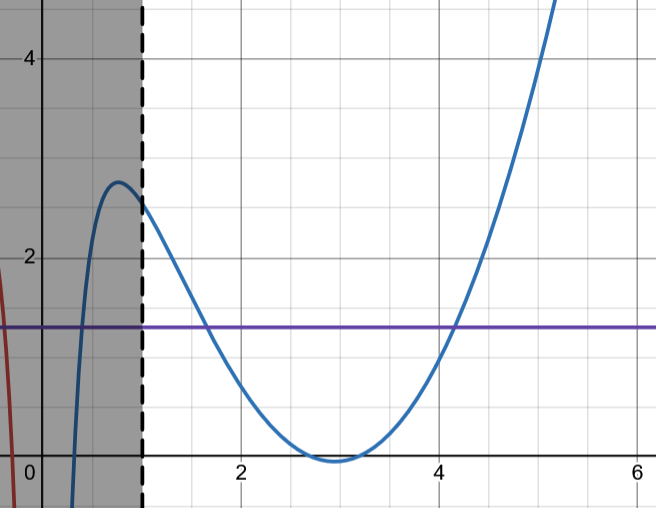
So for these particular parameters there are 2 localized P-states.
N-states¶
If :
Decaying but oscillatory wave-function.
By inserting the large- approximation for the N-state wavefunction in the eigenvalue equation, we get an algebraic equation
which can be solved as before.
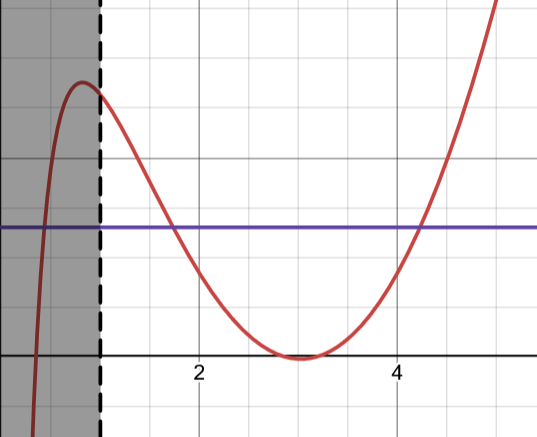
The -states appear at different parameter values than -states. They can even coexist. Check out Desmos!